 Laboratory evaluation revealed a positive SARS-CoV-2 spike antibody test result. On day 15 after the booster vaccination, his palms became severely pruritic, erythematous, and painful. He also reported migratory, intermittent wheals that appeared spontaneously but could be provoked with showering or increased pressure to the skin. The pruritus was most significant at night. Eleven days after the Moderna mRNA-1273 booster dose, severely pruritic papules appeared on his head, extending over the neck and both arms over the next few days. He received both initial Moderna mRNA-1273 vaccinations in early 2021, experiencing transient systemic symptoms, including headache, chills, and fever, but no cutaneous reaction. The patient is a 31-year-old man with no significant medical history who presented with urticaria and pruritic papules following his Moderna mRNA-1273 booster vaccination. The patient will continue the course of treatment with fexofenadine for an additional 4 weeks before any further medication discontinuation trials. Symptoms are managed with regularly scheduled antihistamine therapy however, rebound urticaria recurs within 24 to 28 hours of discontinuing medications. Laboratory data revealed a positive screening for antinuclear antibodies (titer, 1:320 reference value, 6 weeks. She was prescribed 20 mg famotidine twice daily in the event of a breakthrough pruritus, which did not occur while on fexofenadine and vitamin D therapy. The patient subsequently began 180 mg fexofenadine twice daily and 1000 IU vitamin D once daily, which controlled her symptoms. Within 48 hours of discontinuation, a rebound rash appeared. The patient’s urticaria significantly improved with separate trials of diphenhydramine and cetirizine however, the patient discontinued both due to side effects. She intends to receive any recommended booster doses in the future.Ĭase 2 demonstrating dermatographism 3 weeks after administration of the Moderna mRNA-1273 booster dose. On this medication regimen of 300 mg omalizumab subcutaneously once monthly and a daily dose of 180 mg fexofenadine, she received the booster vaccination 8 months after her second dose of the Pfizer BNT126b2 vaccine and did not experience any flare in her symptoms. The patient elected to continue omalizumab 300 mg injections monthly along with daily 180 mg fexofenadine. The dose was subsequently increased to 300 mg with complete clearance. This dosage resulted in partial response. Omalizumab was added at 150 mg subcutaneously for 4 weeks. The patient was continued on H 1 and H 2 blockers. The timing of onset after vaccination, and the dose-response (starting after first dose, exacerbated by second dose) are suggestive of COVID-19 vaccination-induced CSU. Complete blood cell count, liver function panel, and basic metabolic panel data were all within reference ranges. Laboratory data revealed elevated immunoglobulin E levels (288 IU/mL reference value, 0-100 IU/mL) and a positive SARS-CoV-2 spike antibody test result. The dermatopathology report from a punch biopsy indicated interstitial and perivascular inflammatory infiltrate in the dermis with abundant eosinophils consistent with urticaria. The circle indicates the site of the skin biopsy. This material may not be published, broadcast, rewritten, or redistributed.Case 1 at presentation 7 weeks after administration of the second dose of the Pfizer BNT126b2 vaccine. He didn’t recommend taking ibuprofen prior to getting the vaccine by said it was OK to use after the shot is administered. “One or two days of some muscle aches and headaches is a small price to pay to get back to being able to see our families again, prevent hospitalizations, and prevent the deaths that we’ve seen over the past year,” Siedner said. “It’s relatively rare but it’s short-lived and it’s not harmful.”īetween five and 10 out of every one million doses results in a severe allergic reaction, according to the Centers for Disease Control and Prevention. 
“It almost always goes away, so it’s not something that we’re getting too concerned about,” Siedner said. One of the rare side effects of the shot is the formation of a large rash around the injection site, commonly referred to as “COVID arm.” It can be itchy and sore but not dangerous. “This is one of the few times that being older is probably better when it comes to medical issues.” 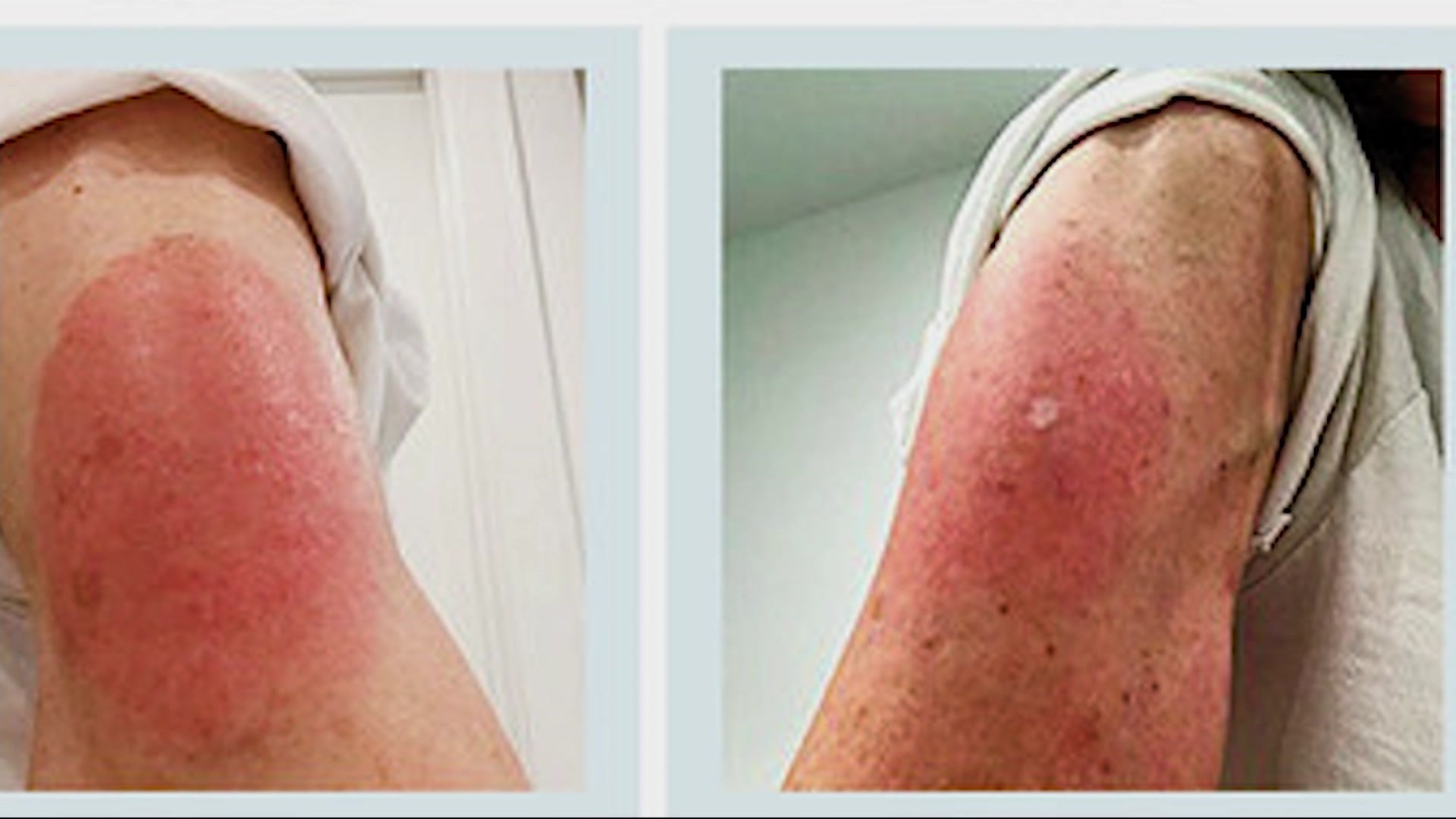
“The older we get, the older our immune system gets, and so the less inflammatory our body will be in response to a virus we’ve seen before when we’re older than when we were younger,” Siedner said. Side effects are actually more common in young people, Siedner added. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
